Quarterly insights: Pharma IT
Enhancing clinical trial diversity

Representing population diversity in trials is important given a review of 167 new molecular entities approved by the FDA between 2008-2013 showed approximately 20% had racial or ethnic differences in safety, efficacy and response. However, trial diversity remains a challenge for drug sponsors.
New guidance from the FDA and others regarding how to broaden eligibility criteria to increase clinical trial enrollment of underrepresented populations provides a roadmap to addressing this challenge. eClinical technology vendors – companies that combine technology, applications, and services to automate clinical trial data collection, management and analysis and to automate other aspects of clinical trials – are providing the tools to make it happen.
We highlight a handful of innovative companies whose software and other technology promote diversity and accessibility in clinical trial enrollment and participation.
Digital biomarkers, developed using digital technologies that make it possible to efficiently collect and manage large volumes of high-quality patient data across dispersed geographies and expansive timelines, are particularly important in achieving diversity and represent an area seeing substantial development, transactions and investment.
TABLE OF CONTENTS
Includes discussion of Dassault/Medidata (Paris: DSY) and ten private companies
- New guidance addresses critical challenge of trial diversity enrollment
- Technology vendors provide tools for the task
- Digital biomarkers promote inclusion and enhance study quality
- Better value for all
- Commercialization and eClinical indices continue recovery
- Pharma IT M&A rebounds in Q4
- Q4 pharma IT private placements continue trend downward
New guidance addresses critical challenge of trial diversity enrollment
COVID-19 has made patient engagement a top priority for the life sciences industry, especially when it comes to recruiting, enrolling and retaining trial participants. Even prior to COVID-19, recruiting and enrolling patients into clinical trials required considerable time and expense. Industry estimates suggest 50% of clinical trials need to extend their recruitment period, resulting in a loss of up to $1.3 million per day for a given drug candidate. 11% of trial sites fail to recruit any patients while 18% of trial participants drop out after enrolling.
Additionally, trial diversity remains a challenge for sponsors despite many trial sites providing medical care for patients of various ethnicities and socioeconomic backgrounds. For example, Black people are about 5% of all patients enrolled in U.S. Food and Drug Administration (FDA) registered clinical trials but represent about 13% of the U.S. population. Adequate representation of population diversity in trials is important given a review of 167 new molecular entities approved by the FDA between 2008-2013 showed approximately 20% had racial or ethnic differences in safety, efficacy and response. Trial diversity becomes particularly important when it comes to COVID-19, which affects patients disproportionately relative to ethnicity, age and economic background.
TABLE 1: Minority underrepresentation in clinical trials
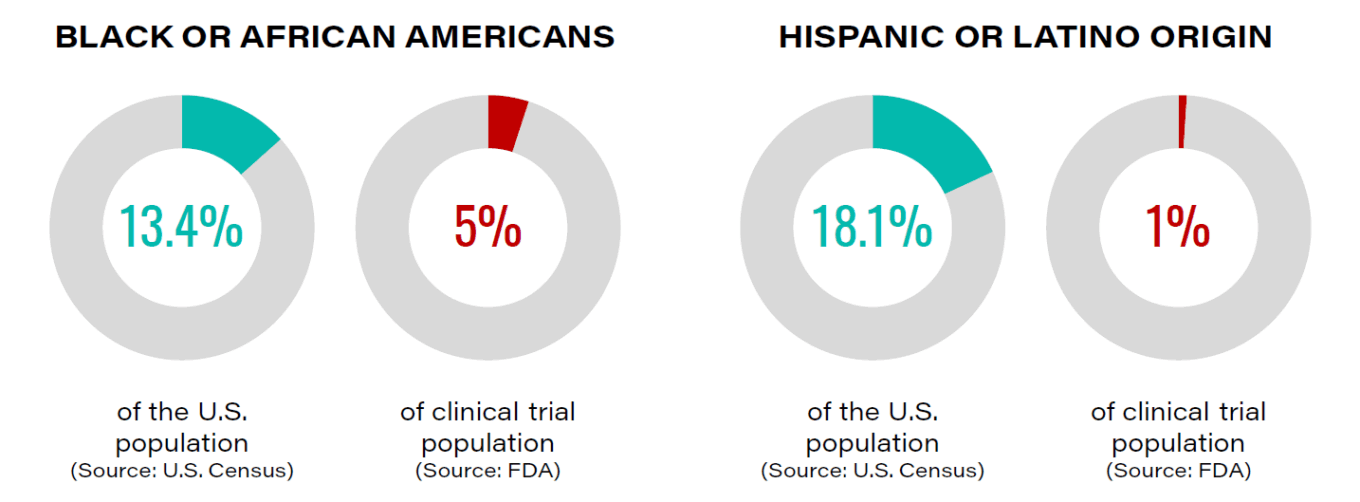
Source: Clinical Research Pathways.
In November, the FDA released new industry guidance titled “Enhancing the Diversity of Clinical Trial Populations – Eligibility Criteria, Enrollment Practices, and Trial Designs.” The guidance documents provide sponsors with a framework on how to broaden eligibility criteria to increase trial enrollment of underrepresented populations. In addition to the FDA, the Pharmaceutical Research and Manufacturers of America (PhRMA) recently published its first-ever industrywide principles on clinical trial diversity, a new chapter in the already existing “Principles on Conduct of Clinical Trials & Communication of Clinical Trial Results.”
The new chapter focuses on four key principles: 1) Building Trust and Acknowledging Past Wrongs, 2) Reducing Barriers to Clinical Trial Access, 3) Using Real-World Data to Enhance Information on Diverse Populations Beyond Product Approval, and 4) Enhancing Information About Diversity and Inclusion in Clinical Trial Participation.
TABLE 2: Model pathway for trial enrollment process
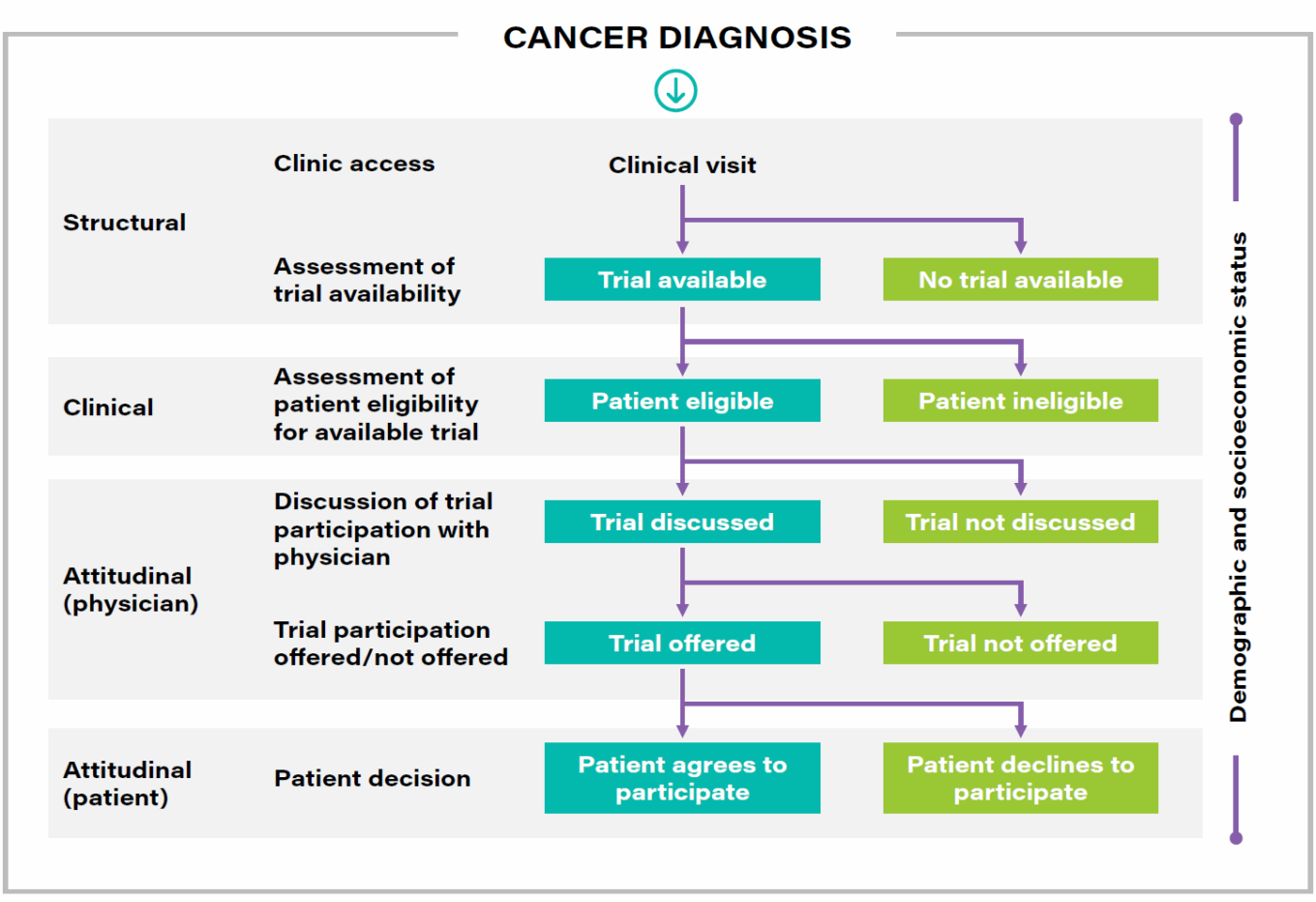
Source: www.fightcancer.org
Technology vendors provide tools for the task
Understanding that proactive patient-centric strategies are needed to identify and enroll suitable patients expected to benefit from a drug’s approval, eClinical technology vendors – companies that combine technology, applications, and services to automate clinical trial data collection, management and analysis and to automate other aspects of clinical trials – are focused on making sure their solutions and services promote diversity and accessibility.
For example, one innovative company providing a differentiated patient engagement solution aimed at loosening the bottleneck of recruitment and enrollment is Ripple Science. Ripple’s CRM-based platform for starting up studies transforms site-level data into patient profiles to help ensure enrollment and retention. In July, Ripple partnered with Lakeside Life Science to quickly recruit and track patients from small, underserved communities to clinically validate Medsential’s point-of-care COVID-19 antibody test.
RxE2 and Matrix Medical Network are two other notable companies we recently spoke with that are using unique engagement models to overcome logistical challenges to trial accessibility and diversity. RxE2 focuses on leveraging the close relationship between patients and pharmacists to improve trial recruitment, retention and outcomes by having pharmacies be trial sites.
According to the company’s CEO, Gerald Finken, pharmacies and pharmacists are significant untapped resources for trial sponsors given 90% of the U.S. population lives within two miles of a pharmacy. Finken noted pharmacies are ideally situated and filled with pharmacists whose skills and expertise make them ideal healthcare professionals to dispense investigational drugs and help ensure medication adherence and compliance. In June, the company partnered with Thrifty White Pharmacy, whose central fill pharmacy is accredited and licensed in all 50 states, to provide sponsors with expanded access to diverse patient populations who may be geographically excluded by the traditional trial site model.
Matrix Medical Network leverages its network of more than 3,000 clinicians and its fleet of mobile health clinics to remove geographic barriers by providing mobile trial access to high-risk and vulnerable populations. In October, the company announced as part of Operation Warp Speed and in collaboration with Fred Hutchinson Cancer Research Center it would provide 20 academic medical centers with mobile health clinics to increase access to the investigational COVID-19 vaccine trials with a focus on those populations most affected by the virus.
Digital biomarkers promote inclusion and enhance study quality
Digital biomarkers are patient-generated physiological and behavioral measures collected through connected digital tools (hardware and software) that can be used to explain diseases, influence fitness behavior, predict drug response and anticipate health-related outcomes. The emergence of digital technologies including smartphones and connected wearables like the Apple Watch provides clinical trial sponsors with the means to generate significant amounts of patient-level data measured at frequent intervals between site visits. These technologies can be applied well beyond the duration of the trial itself, spanning the product life cycle from research and development to product launch to disease management and providing opportunities to develop novel endpoints and supplemental data that may improve the case for therapies’ regulatory and reimbursement approvals.
TABLE 3: COVID-19 decompensation index
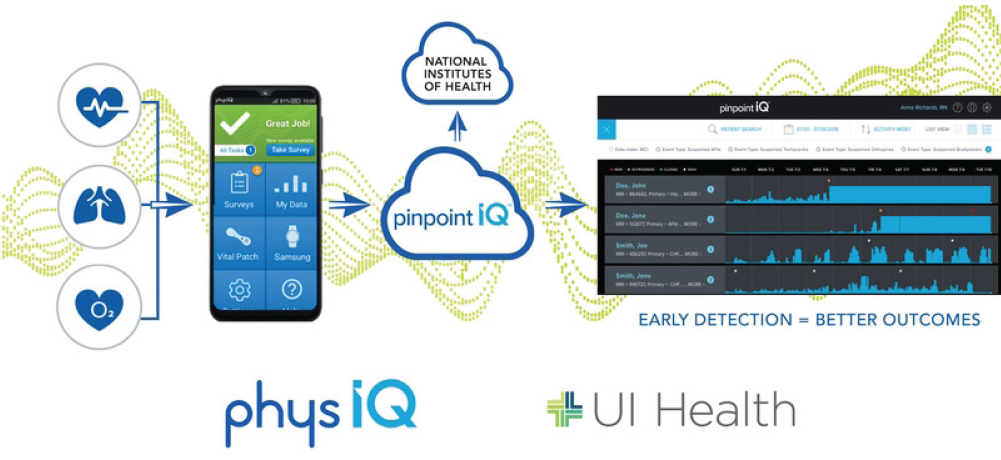
Source: PhysIQ.
As COVID-19 grinds on, demand for digital biomarkers in clinical research has been robust as drug sponsors and clinical research organizations increasingly turn to digital technology vendors for tools that enable them to get trials up and running, keep trials going, and keep patients safe despite the pandemic’s disruptions. Further, the ubiquity, ease of use, relatively low cost, and broad connectivity of the underlying smartphones, other connected devices, and networks that collect digital biomarkers make it possible for bigger, more underrepresented and more remote populations to take part in clinical trials. We believe this creates new opportunities to increase trial diversity and accessibility with digital biomarkers.
For example, in September the National Cancer Institute and the National Institute of Biomedical Imaging and Bioengineering of the National Institutes of Health awarded PhysIQ a contract to develop an artificial-intelligence-based COVID-19 Decompensation Index (CDI) digital biomarker to address the rapid decline of high-risk COVID-19 patients. The study is designed to capture data from a large, diverse population to investigate CDI performance differences among subgroups based on sex, gender, racial and ethnic characteristics.
In another example, Brain Baseline is developing digital biomarkers for Parkinson’s disease through the Watch-PD clinical trial sponsored by the University of Rochester in collaboration with Biogen and Takeda. In our view, the observational study’s hybrid design (in-clinic and at-home) allows for more representative populations to be included, which according to the FDA’s Patient Engagement Advisory Committee is important as different populations who suffer from Parkinson’s disease may exhibit different symptoms. The study is expected to be completed by May 2022. Once validated, the biomarkers are expected to be used to help assess other movement and mobility disorders.
Overall, the market for digital-biomarker-enabled companies has been strong, encouraging more companies to consolidate or raise capital. This is evident in a recent wave of M&A and financing activity in the space, including Medidata’s (a subsidiary of Dassault Systèmes, Paris: DSY) recent acquisition of MC10’s digital biomarker business, ArchiMed’s acquisition of Actigraph, ERT’s acquisition of APDM and recent merger with Bioclinica, AliveCor’s $65 million Series E financing, Evidation Health’s $45 million Series D financing, and Koneska Health’s $16 million Series B financing.
Better value for all
By providing tools that enable therapeutic companies to efficiently understand and optimize how their products affect all possible target populations, eClinical technology companies help life science innovations reach every potential beneficiary, maximizing the value society and the economy derive from our rapidly advancing understanding of human biology.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
