Quarterly insights: Process technology
A fungus among us, and that could be a good thing

We explore in detail an example of the theme we discussed in our last Process Technology Quarterly Insights: how scientific wars and races stimulate innovation and create opportunities for new technologies and market entrants throughout the economy, including pharmaceuticals. In our example, Dyadic International (DYAI), an innovator in bioproduction technology, stands to see uptake of its technology platform for biopharmaceutical production accelerated by the urgency of developing and producing COVID-19 vaccines and treatments.
Dyadic’s C1-based expression system for drug development and production, already battle tested in the industrial markets, has the potential to disrupt the market for bioproduction of human therapeutics by enabling much more productive, lower-cost bioproduction processes.
But while C1 stacks up well in terms of speed, efficiency, productivity, and cost relative to other expression systems used in drug development and production, it lacks a regulatory and safety history in human therapeutics. Now, with COVID-19 as a catalyst for adoption, Dyadic has a number of partnerships and programs underway, many tied to COVID-19 vaccines and treatments, aimed at establishing C1 in the human therapeutics market.
TABLE OF CONTENTS
Includes discussion of DYAI, MRNA, SNY
- What is an expression platform?
- A little more on C1
- Opportunities to break through
- Transformational potential
- Process technology index continues post-COVID-19 rally
- Process technology M&A activity begins to rebound
- Process technology private investment slows in Q3
Introduction
In our last Process Technology Quarterly Insights report, we provided an overview of pharmaceutical technology and service providers with emphasis on the push for COVID-19 therapeutics. We stated our opinion that wars and scientific races, like the race for COVID-19 vaccines and treatments, stimulate innovation and create opportunities for new technologies and entrants throughout the economy, including in the pharma industry.
In this report we highlight an example: The race for COVID-19 therapeutics is creating new opportunities – and perhaps the potential to be a major technological disruptor – for an innovator in bioproduction technology, Dyadic International.
Dyadic has a long history of developing and commercializing its C1 fungal expression platform. Over time, it has invested several hundred million dollars in development. Dyadic successfully commercialized C1 for industrial applications and ultimately sold the technology to Dupont for $75 million. In our view, the high-volume, low-price nature of industrial biotech makes it a more technically challenging market than healthcare, a low-volume, high-price market.
Biotech pioneers Genentech and Amgen flirted with industrial biotech initially but quickly perceived the human therapeutics market as much more attractive, and the rest is history. But given Dyadic’s C1 platform has been successfully battle tested in the industrial markets, we think it now has the potential to disrupt the market for bioproduction of human therapeutics by enabling much more productive, lower-cost bioproduction processes. Dyadic has a number of partnerships and programs underway aimed at establishing C1 in the human therapeutics market.
TABLE 1: The 20-year evolution of C1 platform technology
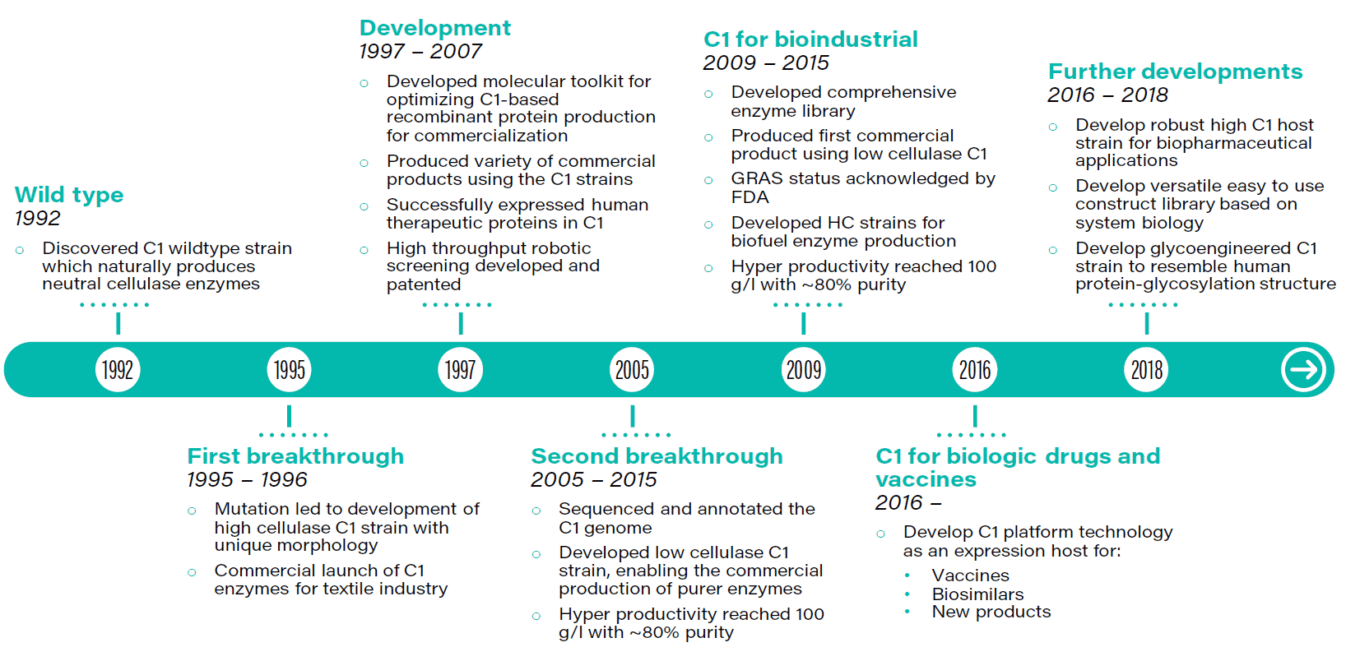
Source: Dyadic.
What is an expression platform?
One of the key elements of biotherapeutic production is the host or expression system. In simple words, it is a cell line used to produce a drug, which is typically a protein (or possibly a metabolite) with therapeutic properties. There are a number of established expression platforms based on cells of organisms including bacteria (Escherichia coli), yeast (a type of fungus, such as Saccharomyces cerevisiae), insects, mammals (Chinese hamster ovary, known as CHO), and humans (Moderna’s mRNA system).
In addition, there are non-yeast fungal cell lines, one of which is Thermothelomyces heterothallica, or C1, a mutant filamentous fungus, which Dyadic has developed as a robust platform for protein and metabolite production and which has been successful in industrial biotech applications.
Each type of cell has unique attributes, requirements (including nutrients and other process conditions) and biochemical machinery for producing proteins. Therefore any particular expression system may be more or less well suited for producing a desired product. In many cases, the toughest challenge is to first keep the cells alive and then highly productive at commercial scale.
Other challenges include purification: If the platform produces proteins at low concentrations or proteins mixed with other hard-to-separate chemicals or doesn’t secrete the desired proteins, purification can be difficult and costly. Here we list a few of the most desired attributes:
- High expression levels: The cells should produce a lot of the desired protein per milliliter of fermentation broth, avoiding inhibition and other limiting effects.
- High expression rates: The cells should process the desired protein quickly.
- Easy processing: This encompasses a number of considerations. For example, the process should not require unusual conditions, such as extreme temperature or pH, have too high or too low viscosity or require expensive input feeds. It should work well at both large and small scales, and it should be easy to separate the desired protein from other chemicals in the output.
- Easy cell line programming and engineering: It should be easy to genetically modify the cell line to make a variety of desired proteins and not make undesired byproducts such as proteases.
- Safe: The cell line should have a long and safe regulatory history for products used in humans, typically meaning the FDA is familiar with the cell line and accepts it.
TABLE 2: What is C1? Thermothelomyces Heterothallica
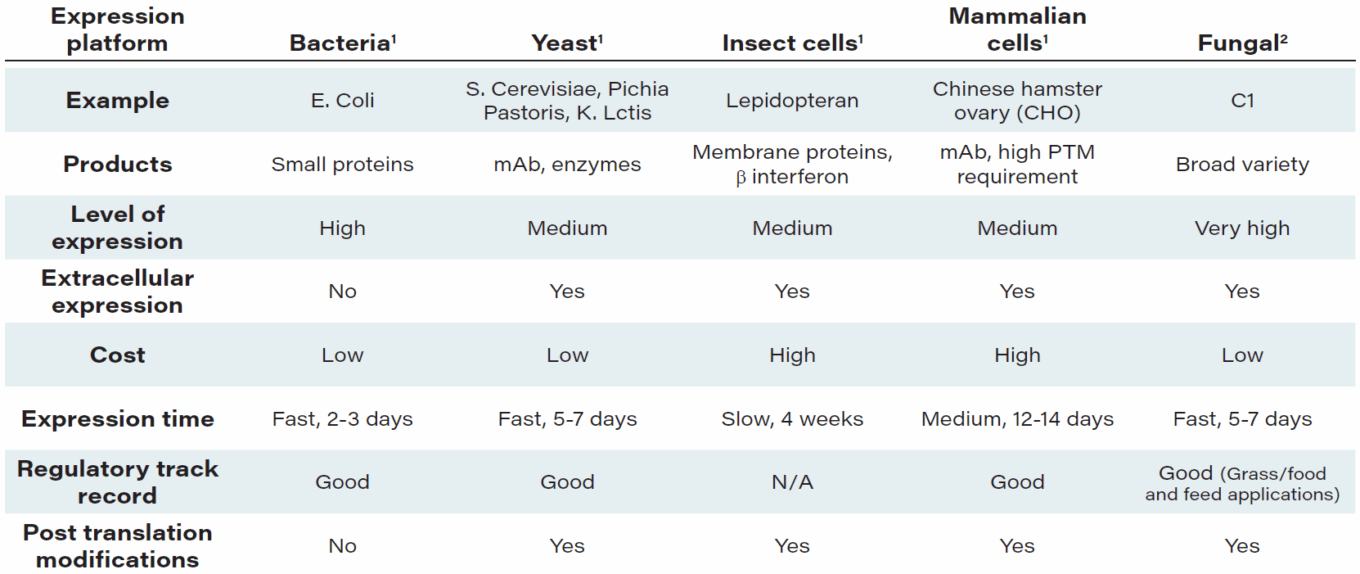
Source: Dyadic.
Notes: 1. Niazi, Sarfaraz K. (2016) Biosimilars and Interchangeable Biologics Tactical Elements 2. Dyadic data and assessment
The first four bullets really boil down to driving the manufacturing cost of the drug lower. Simply put, if you can rapidly develop a cell line that can generate product quickly and in high concentrations, all costs and timelines shrink. Instead of months or years to first product, maybe it’s weeks. Instead of a 10,000-liter stainless-steel reactor, maybe you can use a 50-liter single-use reactor. It’s on these types of measures that Dyadic’s C1 platform excels.
For example, in a recent update Dyadic noted its C1 platform demonstrated three to four times the productivity of CHO cells in producing COVID-19 antibody cocktails. It also reported its C1 platform expressed receptor binding domain antigen of the COVID-19 coronavirus spike protein at a relatively high 3 grams per liter as part of the antibody vaccine efforts, and it expects 10 animal trials to be completed by year end.
But for the moment, at least, we believe the other lines have the clear advantage on the last bullet. You don’t get fired for choosing a CHO system, which is well established in human biopharmaceutical production, for your monoclonal antibody system. In this respect, we see some interesting similarities between Moderna’s (MRNA) mRNA technology platform and Dyadic’s C1 fungal system since their platforms have no prior approvals with the FDA. It is in their lack of safety history and regulatory history where C1 and Moderna’s mRNA systems fall short.
While we can debate why no C1 products are yet approved for human health, the important point is that the pandemic has created a sense of urgency, a willingness to invest in innovative solutions, and increased regulatory flexibility that are likely to help these platforms clear the human safety hurdle with human clinical trials in the near term. We don’t even think C1 needs to be commercially successful with a COVID-19 therapy, as Dyadic has numerous shots on goal in other therapeutics programs; however, it does need a successful human trial. (We note Moderna, with its high-profile mRNA COVID-19 vaccine candidate, has a current market cap of about $27 billion versus Dyadic’s about $200 million.)
A little more on C1
Based on industrial applications, C1 has been shown to be a hyperproductive cell line with good processing parameters. Key processing parameters and characteristics that make C1 attractive include:
- Process temperature of 20-45 Celcius, pH of 5-8
- Uses a simple sugar feed input
- Has a unique multicellular morphology, or structure, that increases productivity and rheology in the reactor, an area where many other filamentous fungal organisms fall short
- Produces no endotoxin or viral contamination
- Can produce more human-like glycosylation patterns and protein folding relative to other lower-organism systems
- Most proteins are secreted, simplifying processing and purification
- Has been demonstrated to be scalable from lab to commercial volumes
Opportunities to break through
Dyadic’s C1 platform is being used in numerous partnerships and programs aimed at applications in human health. These include partnerships with:
- VTT Technical Research Centre of Finland to develop a more efficient and lower-cost alternative to produce therapeutic proteins, vaccines and metabolites based on C1, including a new production method for a COVID-19 vaccine;
- the Serum Institute of India, a large vaccine manufacturer, to develop 12 antibody products and vaccines;
- Sanofi (SNY), to express all seven of Sanofi’s proteins and vaccines (half have exceeded target expression levels, and Dyadic is waiting for Sanofi to decide next steps);
- Frederick National Laboratory, to produce vaccine candidates to be used by the Vaccine Research Center;
- the Israel Institute for Biological Research, to explore the potential of Dyadic’s C1 platform to express gene sequences and targets developed by the institute into both a vaccine candidate and monoclonal antibodies that may help combat COVID-19;
- Erasmus Medical Center, Utrecht University and the University of Veterinary Medicine Hannover, to develop a COVID-19 vaccine candidate; and
- Ufovax to begin protein expression based on Ufovax’s gene sequences for potential COVID-19 and HIV vaccine candidates.
Dyadic has other partnerships and programs in animal health, including a partnership with the Zoonotic Anticipation Preparedness Initiative (ZAPI). ZAPI is a research and development program sponsored by the EU aiming to build a platform for rapidly developing and producing vaccines and protocols and to fast-track registration of developed products to combat epidemic zoonotic diseases that have the potential to affect humans. Two antigens have been developed and tested in animals, one for Schmallenberg virus and one for Rift Valley fever virus. Dyadic is also working with four of the top global animal health companies.
Transformational potential
As we explored in our June Process Technology Quarterly Insights report, major societal and economic stress events like the COVID-19 pandemic often transform industries and technology. The potential for Dyadic’s C1 platform, with its attractive cost and speed profile that makes it highly compelling in the context of the pandemic, to clear the human safety hurdle and thereby achieve widespread acceptance and adoption in human biopharmaceutical production, is an example of such transformational possibilities.

Request full report
To access the full report, please provide your contact information in the form below. Thank you for your interest in First Analysis research.
